Shengju Wu, Fengting Li, Yinan Wu, Ran Xu and Guangtao Li, Chem. The determining factors of the formation of a precipitate can vary. Preparation of novel poly(vinyl alcohol)/SiO 2 composite nanofiber membranes with mesostructure and their application for removal of Cu 2+ from waste water Precipitates are insoluble ionic solid products of a reaction, formed when certain cations and anions combine in an aqueous solution. Commun., 2009ĭendronized iron oxide nanoparticles as contrast agents for MRIīrice Basly, Delphine Felder-Flesch, Pascal Perriat, Claire Billotey, Jacqueline Taleb, Geneviève Pourroy and Sylvie Begin-Colin, Chem. Theivanayagam Muraliganth, Arumugam Vadivel Murugan and Arumugam Manthiram, Chem. Commun., 2009įacile synthesis of carbon-decorated single-crystalline Fe 3O 4 nanowires and their application as high performance anode in lithium ion batteries Hydrophilic multi-walled carbon nanotubes decorated with magnetite nanoparticles as lymphatic targeted drug delivery vehiclesĭong Yang, Feng Yang, Jianhua Hu, Jiang Long, Changchun Wang, Deliang Fu and Quanxing Ni, Chem. Analogously, in medicine, coprecipitation is specifically the precipitation of an unbound 'antigen along with an antigen-antibody complex'. BioSyst., 2009Įlucidating the genesis of Bi 2MoO 6 catalyst by combination of synchrotron radiation experiments and Raman scatteringĬhanapa Kongmark, Vladimir Martis, Annick Rubbens, Caroline Pirovano, Axel Löfberg, Gopinathan Sankar, Elisabeth Bordes-Richard, Rose-Noëlle Vannier and Wouter Van Beek, Chem. In chemistry, coprecipitation (CPT) or co-precipitation is the carrying down by a precipitate of substances normally soluble under the conditions employed. A precipitation reaction is a reaction between two reactants (most commonly aqueous solutions of ionic compounds) in which a solid (precipitate) forms. Phosphoproteomics-finally fulfilling the promise? Sankaran Murugesan and Vaidyanathan (Ravi) Subramanian, Chem. Thesaurus: Washington Irving Bracebridge Hall, or The Humourists. a journey, or a conflict Richard Glover Back to his sight Francis Bacon The Essayes OCLC if they be stout and daring, it may their designs, and prove dangerous ( transitive) throw height. Robust synthesis of bismuth titanate pyrochlore nanorods and their photocatalytic applications ( transitive) To make something happen suddenly and quickly. The most common examples of precipitation are Rain, Snowfall, hail, sleet, dew etc.More about the RSC Chemical Methods Ontology (CMO)įabrication of living cellosomes of rod-like and rhombohedral morphologies based on magnetically responsive templates Question: What are examples of precipitation occur in nature?Īnswer: The precipitation occurs in nature, Type and size of ions, the concentration of an aqueous solution, pH of the solution, solubility etc. Question: What are the factors that affect the precipitation reaction?Īnswer: Factors affecting precipitation reaction are, Definition of PRECIPITATE (verb): make something suddenly happen or start existing solid substance: become separate from liquid fall or be thrown. 
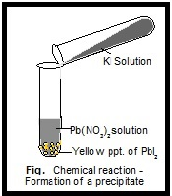
It is a type of double displacement reaction. It is used to check out elements present in the solution. The precipitate is shown by the down arrow in the chemical reaction. Given enough time, the solid will settle down to the. The chemical reaction in which two ions combined to form one of the insoluble products in an aqueous solution that precipitated. 5 Notes on Precipitates When we mix two liquid chemicals and get a solid, we call that solid a precipitate. Frequently Asked Questions: Question: What is a precipitation reaction? It affects by the type and size of ions, the concentration of an aqueous solution, pH of the solution, solubility etc.Acting with or marked by excessive haste and lack of due deliberation. These insoluble salts formed in precipitation reactions are called precipitates. Moving rapidly and heedlessly speeding headlong. The term ‘precipitation reaction’ can be defined as a chemical reaction occurring in an aqueous solution where two ionic bonds combine, resulting in the formation of an insoluble salt. It is a type of double displacement reaction. Chemistry To be separated from a solution as a solid.The precipitate is shown by the down arrow in a reaction. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
